Search

Painful monthly penicillin injections to treat and prevent Rheumatic Heart Disease (RHD) could be replaced with a longer-acting implant, a recent study has found.
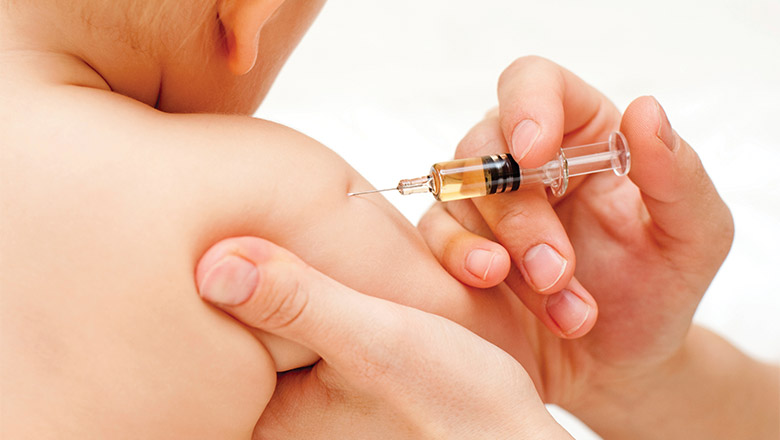
Parents of babies in Perth and Adelaide are being urged to take part in a landmark study to examine the best ways to keep their child safe from influenza this winter, amid a surge in serious infections.

People living with hepatitis C in WA are being urged to take part in a new project, aimed at encouraging the take-up of treatment.

The Kids Research Institute Australia researchers will lead a new national clinical trial (COSI-2) to determine whether topical coconut oil can reduce late onset sepsis in extremely preterm infants.

The Deborah Lehmann Research Award in Paediatric Infectious Disease Research is a funding mechanism to support the training and development of early- to mid-career researchers (EMCR) or Higher Degree by Research (HDR) students who are nationals from the Pacific Region working in or outside their hom

More children across Australia are being vaccinated against the flu since funding was expanded and access widened under the National Immunisation Program
Thank you to our Sponsors Contact us If you'd like to get in touch, please contact Marie Nadal-Sims by phone or email. Phone: (08) 6319 1001 Email:
Contact us If you'd like to get in touch, please contact Marie Nadal-Sims by phone or email. Phone: (08) 6319 1001 Email: IICPerth@thekids.org.au All
Contact us If you'd like to get in touch, please contact Marie Nadal-Sims by phone or email. Phone: (08) 6319 1001 Email: IICPerth@thekids.org.au All

Wrap up of the people, projects and updates from 2017 in the Vaccine Trials Group research area, and Dr Peter Richmond.
